Revolutionizing IVF: UC San Diego Researchers Develop Non-Invasive Embryo Quality Prediction Method
January 17, 2024
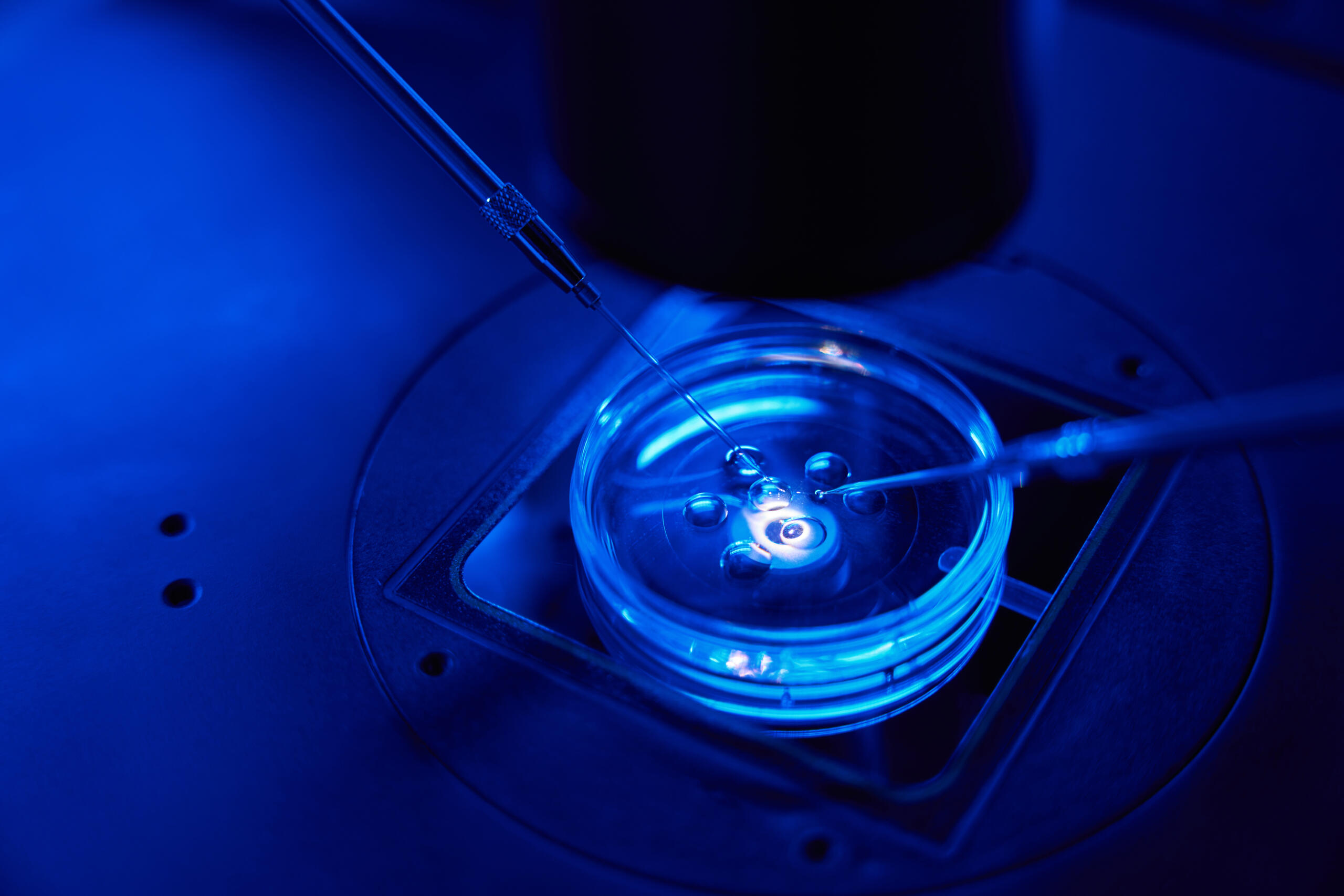
Researchers at UC San Diego School of Medicine have developed a new method to improve the prediction of embryo quality in IVF, potentially increasing the chances of successful pregnancies. IVF, a beacon of hope for those facing infertility, involves fertilizing eggs in a lab and implanting embryos in the uterus. However, the process is often challenging, with multiple rounds of treatment being shared due to difficulty in predicting which embryos will lead to successful pregnancies.
The innovative approach, detailed in Cell Genomics, involves analyzing extracellular RNA (exRNA) in the liquid media where embryos are cultured. This non-invasive technique, developed by senior scientist H. Irene Su and colleagues, contrasts with current methods that assess embryos based on morphology or cell biopsies, which have limitations in predicting healthy baby development.
The study involved analyzing culture media from fertilized eggs, mapping exRNA to the human genome, and identifying distinct genetic profiles indicative of different embryonic stages. This led to the creation of an atlas categorizing the stages from egg to blastocyst, the point ready for implantation. The method also uses machine learning to grade embryo quality, correlating well with traditional morphology-based assessments.
Victoria Jiang, a reproductive endocrinology and infertility fellow at Massachusetts General Hospital who was not involved in the study, highlights the potential of this advancement. It could replace more invasive, expensive, and skill-dependent biopsy methods, ultimately improving financial and pregnancy outcomes.
However, the study faced challenges, such as analyzing shared media from grouped embryos, making it difficult to determine the exRNA origin. Further research is required to confirm the efficacy of this technique for individual embryo selection and to understand the role of identified genes in arrested embryos. While not replacing embryologists, this method could become a valuable tool in embryo selection and understanding human embryo development.
To read more, click here.
[Source: STAT, January 17th, 2024]






