Olympus Asks Healthcare Providers to Stop Using Its Insufflator Units Due to Over-Inflation Reports; FDA Issues Class I Recall
October 31, 2023
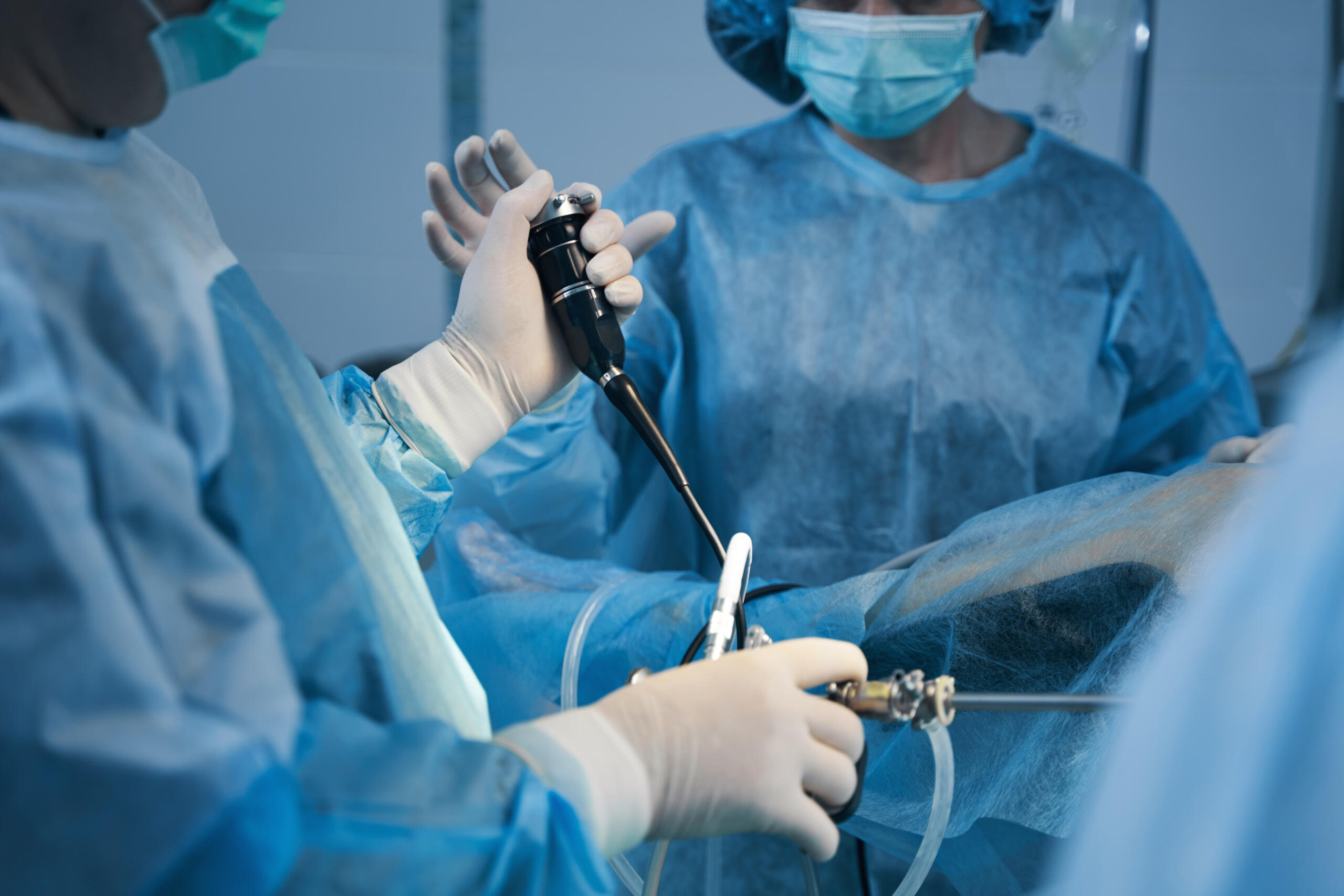
Olympus, a healthcare equipment company, has asked healthcare providers to stop using its UHI-4 insufflator unit, used for laparoscopic surgeries, due to reports of over-inflation potentially causing patient harm. This system works by pumping carbon dioxide gas into the abdomen to create space for performing minimally invasive procedures. The FDA has labeled this issue a class, I recall, given the severity of 21 malfunctioning incidents reported, including ten severe injuries and one death. Olympus has not asked for the units to be returned; instead, they should be quarantined and carefully marked to avoid accidental use. The company is working on additional instructions and is identifying patient subgroups that might be more at risk. Olympus has been issued three FDA warning letters in five months over its endoscope manufacturing following an inspection of its Tokyo facilities.
To read more, click here.
[Source: Fierce Biotech, October 31st, 2023]






